Launch of branded product into mental health market
The Challenge
Launch of a branded prolonged release antipsychotic drug into the market
Many generic scripts in the market written generically and the perception that generic prescribing was the lowest cost option
Many CCG’s saw the best cost saving scheme was to move from MR preparations to IR preparations
Push back from pharmacists and dispensing doctors who would loose profit by moving to branded generic
Moving away from generic prescribing meant that had to be confidence in supply chain as the brand would have to be readily available
Many competitors introduced into the market at the same time.
Our Approach
Target CCGs with cost and patient benefit message
In-house data sources allows bespoke CCG materials.
Specialist savings information, supply chain and pricing information.
Provision of review service to support prescribing changes.
Data systems changed to reflect product of choice
Implementation of Dispensing Doctor schemes.
Timed localised wholesale and retail campaign in CCGs where on formulary
Targeted rebates as required
Results
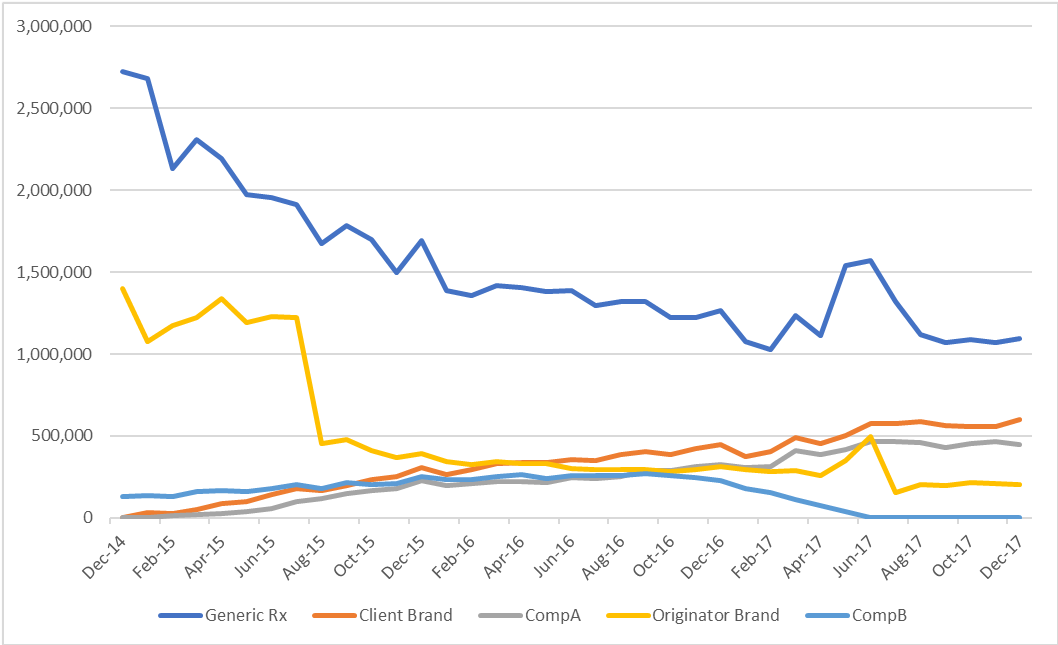
Dominant market share gained within 18 months, taking product to brand leader position.
Gaining 121 CCG formulary inclusions.
Exceeded client growth expectations by 21%.
• Brand leader within 18 months
• Brand originator price reduction did not slow growth
• Competitors failed to match growth even with better price offer
Launch of preservative free eye drop range
The Challenge
To launch a range of preservative free eye drops in a multi dose dropper bottles to the primary care market
Competing with less expensive preserved drops
Preservative free historically meant unit dose vials- patient education
Primary care did not initiate treatment or make switches without secondary care endorsement
Our Approach
To target major secondary care eye units and engage in peer to peer discussions to gain endorsements, regarding patient and environmental benefits
Gaining hospital formularies and utilizing this endorsement at primary care level. Utilizing advocates in one region to influence others
Establishing a need for preservative free product in a multi dose bottle Vs UDV’s
Development of materials for patients on use of dropper bottles to aid compliance and acceptance of the delivery device
Tele-marketing campaign to all CCGs
Pull through by patient reviews and PCN pharmacist involvement
Sponsorship of society meetings to gain contacts and create awareness of the brand
Results
Defining the company previously not know in the ophthalmology field as a major player in this arena
Gaining all key eye hospital formularies. and endorsement
Gaining 201 CCG formulary inclusions
Gaining 51% market share
Over 50% market share after 21 months in the market
Key eye centres ordering directly after 12 months in the market
Stroke review service for pharma client
The Challenge
Launch Product in a therapy area where there were already 3 other leading competitor brands
Large Pharma companies had already secured a stronghold
Obtaining both secondary and primary care formulary inclusion
Establishing first line positioning
Develop a robust mechanism for PCO’s to change from the ‘big 3’ brands to the client’s product
Covid 19 – issues with access to primary care sites
NHS need to move patients from Warfarin to a DOAC
How to become this DOAC of choice
Our Approach
Target CCGs with cost and patient benefit message
Interventions to reduce incident of ischaemic stroke
Optimise dose and treatment plans
Enhance patient monitoring and reduce adverse effects
Bespoke CCG materials from inhouse data
Both onsite and remote access available
Data systems changed to reflect product of choice
Results
Results to date – service is still active
Product of choice in reviewed CCG’s
400% growth in reviewed CCG’s (Qtr vs Qtr)
Moved from 4th to 2nd place market position in reviewed CCGs – Rx Volume
Effect of Rx review in Completed CCG’s
Rx value
Effect of Rx review in individual CCG’s Rx Value